Aquacover
Can You Drink Water from a Water Softener?
Can you drink water from a water softener? Yes, is the answer, but it depends. Most people can safely drink softened water with no ill effect; the only noticeable difference is in the taste: some people like it, others don’t. Some people with skin conditions, such as eczema, have also reported an improvement in their condition using soft water for washing but evidence is limited.
However, higher quantities of sodium (used in the ion exchange process of removing calcium and magnesium) may not be suitable for babies, or people with cardiovascular problems as it can increase hypertension, and it’s for these reasons the Drinking Water Inspectorate and local water companies advise, where water softeners have been installed, you keep a separate supply for drinking that has not passed through the softener.
Read on to find out more about soft and hard water, drinking water standards, calcium, magnesium and sodium in our water, and what might prevent you from drinking softened water.
What is Soft Water?
First off, what is soft water?
As water falls from the sky and lands on the ground it begins a lengthy journey through the water table, reservoirs, rivers and streams, over fields, and through rocks and soils to eventually end up in our houses as the essential liquid we all use in our daily lives.
Depending on the journey it takes water may collect and dissolve various minerals as it percolates through rocks and soils.
Naturally ‘soft’ water has run over non-porous rocks such as granite, slate and marble and dissolves few minerals.
If, however, during its course water percolates through more porous rocks such as chalk, limestone or gypsum it will dissolve a certain amount of calcium and magnesium. It is these minerals, especially calcium carbonates, which make water ‘hard’.
Hence some areas of the country are soft water areas and some are hard water areas and this entirely depends on the local geology.
Water hardness is the traditional measure of the capacity of water to react with soap, hard water requiring considerably more soap to produce a lather, and expressed as the equivalent amount of calcium carbonate in parts per million (mg/l).
The World Health Organisation (WHO) states general guidelines on water hardness:
“Water containing calcium carbonate at concentrations below 60 mg/l is generally considered as soft; 60–120 mg/l, moderately hard; 120–180 mg/l, hard; and more than 180 mg/l, very hard.”
This is echoed by the United States Geological Survey (USGS):
“General guidelines for classification of waters are: 0 to 60 mg/L (milligrams per liter) as calcium carbonate is classified as soft; 61 to 120 mg/L as moderately hard; 121 to 180 mg/L as hard; and more than 180 mg/L as very hard.”
Water companies in the UK however differ slightly in their grading but the overall bands are very similar. Southern Water offers the following guidelines:
- Soft water contains less than 100mg of calcium carbonate per litre
- Moderately hard water contains between 100 and 200mg of calcium carbonate per litre
- Hard water contains between 200 and 300mg of calcium carbonate per litre
- Very hard water contains more than 300mg of calcium carbonate per litre
While, Severn Trent Water says:
| Hardness as CaCO³ | Hardness Level |
|---|---|
| 0-50 | Soft |
| 51-100 | Moderately soft |
| 101-150 | Slightly hard |
| 151-200 | Moderately hard |
| 201-300 | Hard |
| Over 300 | Very hard |
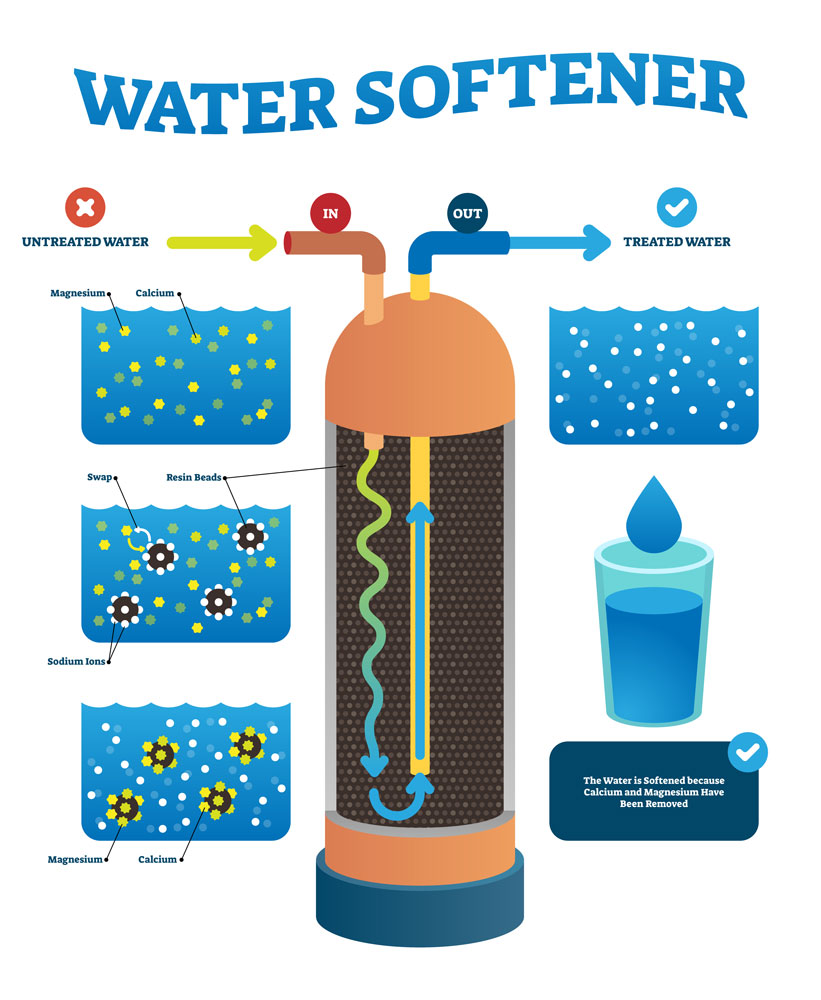
Where water has been through a water softening process such as in a domestic water softener system, it’s correctly known as ‘softened’ water.
Do You Have Hard Water?
Hard water can cause problems with domestic and commercial water systems including heating elements, pipes and boilers making them highly inefficient. Call us on 01773 521437.
Drinking Water Quality Standards

Drinking water quality in England and Wales is monitored by the Drinking Water Inspectorate (DWI), a division of the Department of Environment, Food and Rural Affairs (DEFRA), whose job it is to “provide independent reassurance that water supplies in England and Wales are safe and drinking water quality is acceptable to consumers”. Scotland has the Drinking Water Quality Regulator (DWQR) and Northern Ireland the Drinking Water Inspectorate – Northern Ireland (DWINI).
According to the Inspectorate, “Where water companies artificially soften water before putting it into supply, it is recommended that they maintain a minimum total hardness of 150 mg/l (as calcium carbonate). This is because there is some limited evidence of a relationship between water hardness and cardiovascular health which may be related to the beneficial properties of magnesium and calcium in the diet.”
Some water companies however impose upper limits on water hardness but this is more for practical reasons than health. As Severn Trent Water says: “… hardness should not normally exceed 500 mg/l for household use because some individuals can taste the minerals and it can cause a high level of scaling in pipes.”
What is the Problem with Hard Water?
The term ‘hard water’ may conjure up images of good vs bad or right vs wrong but it’s not the case at all: there are in fact several advantages to hard water, and a few disadvantages.
Advantages of Hard Water
Both calcium and magnesium are essential for our day-to-day metabolism and, as seen, are often naturally present in water. These minerals build and strengthen our bones, teeth and muscles, and aid the nervous and digestive systems among many benefits.
Severn Trent Water asks whether hard water is harmful to health and replies “No, not even very hard water … Some studies have shown that hard water may contribute to a worsening of existing eczema in some individuals. The reason for this is uncertain and the evidence is very limited. However, if a water softener is installed for this reason, the authorities still recommend that a drinking water tap that is not softened is retained.”
The taste of hard water is also often cited in the debate. Some people prefer the taste of hard water but this is clearly subjective and down to age, genetics and experience. Some say softened water tastes “odd” while others say hard water gives them a “chalky mouthfeel”.
According to the WHO “The taste threshold for the calcium ion is in the range 100–300 mg/l, depending on the associated anion, but higher concentrations are acceptable to consumers”. So even the quantity of calcium can vary within an acceptable range which in turn influences taste.
Disadvantages of Hard Water
Hard water can be extremely damaging to domestic, commercial and industrial appliances though and this is where we pay close attention as it produces a substance known as limescale (or simply scale) which in turn reduces the appliance’s efficiency and lifespan, and the efficacy of soaps and detergents resulting in less lather. If left untreated limescale can cause irreparable damage.
Hence, this is where water softeners are used: to reduce limescale water softening equipment artificially softens water through a process known as ion exchange where sodium ions replace the dissolved calcium and magnesium ions.
In an industrial setting limescale can cause damage to cooling towers, commercial premises and boilers.
In a domestic environment hard water can damage washing machines, dishwashers and central heating systems. These are a considerable expense and costly to repair and replace. In addition, hard water can leave ‘tide marks’ on basins, sinks, baths and toilets and a scum on the surface of hot drinks.
But, it is the increased level of sodium that causes debate on whether people should drink softened water or not (see below).
According to the DWI, “Some people with skin conditions, such as eczema, have reported an improvement in their condition from using soft water for washing” but admit data confirming this is limited.
They continue, “If you do install a water softener, it is very important that you make sure that it is correctly installed and you do not soften the water to the tap in your kitchen which is used for drinking and cooking. This is because most water softeners work by replacing the hardness with sodium. Too much sodium can be a problem for premature babies because their kidneys are not good at filtering it out of the blood, and for people who are on a low sodium (low salt) diet. Artificially softened water may also be aggressive to plumbing causing leaching of copper and lead.”
Calcium and Magnesium in Our Diet
In a litre of moderately hard water we might estimate 150 mg of calcium carbonate.
Water isn’t the only source of calcium and magnesium in our diet though so removing them from water, for most people, isn’t going to suddenly cause a deficiency. (Remember, people are more concerned about the increased sodium content especially those with hypertension and babies.) Many foods and drinks are also high in calcium and magnesium:
Calcium
The NHS advises adults aged 19 to 64 need 700mg of calcium a day and you should be able to get all the calcium you need from your daily diet:
- Milk, cheese and other dairy foods
- Green leafy vegetables – such as broccoli, cabbage and okra, but not spinach
- Soya beans
- Tofu
- Soya drinks with added calcium
- Nuts
- Bread and anything made with fortified flour
- Fish where you eat the bones – such as sardines and pilchards
Magnesium
The NHS also advises the daily amount of magnesium you need is:
- 300 mg a day for men (19 to 64 years)
- 270 mg a day for women (19 to 64 years)
You should be able to get all the magnesium you need from your daily diet such as:
- Green leafy vegetables – such as spinach
- Nuts
- Brown rice
- Bread (especially wholegrain)
- Fish
- Meat
- Dairy foods
How much sodium (and salt) is in softened water?
It is difficult to say how much sodium is in softened water as it entirely depends where you live and how much is required.
According to Severn Trent Water, “… softeners can cause high levels of sodium in the water and it is strongly advised by medical experts that you keep a separate supply for drinking that has not passed through the softener.
“Electromagnetic water conditioners are also available. These fit around the pipe and do not come into direct contact with the water. They do not remove the hardness salts, but claim to reduce scale formation. Reports on their performance have been variable, but the water treated by such devices is safe to drink.”
They then ask whether softened water is harmful to which they reply “Normally, no. However, a separate unsoftened tap should be provided for drinking water if an ion-exchange softener is fitted. Do not use artificially softened water in babies feeds or for people on a low sodium diet.”
In this respect, according to the British Heart Foundation (BHF), “Eating too much salt may raise your blood pressure, and having high blood pressure increases your risk of developing coronary heart disease. So to keep your heart healthy it’s important that you don’t eat too much salt each day.”
There is also evidence to suggest increased amounts of sodium may be an issue for toddlers.
Water companies in the UK follow the DWI’s requirements on sodium levels and advise this should be 200 mg/l (0.2g) when it reaches the point of turning on the tap.
For water that needs softening at home we advise for every 100mg/l of calcium carbonate, add 46 mg/l of sodium.
Attention: Despite softening water with sodium don’t think drinking soft water is like taking a huge gulp of sea water: softened water may have a slight salty tinge but it is usually indiscernible. Also, do not to confuse sodium measurements with those of salt.
Sodium (Na) comprises 40% of salt (NaCl) so to convert sodium measurements to salt, multiply the sodium amount by 2.5. For example, 1g of sodium per 100g is 2.5 grams of salt per 100g.
The NHS recommends adults eat no more than 6g of salt a day (2.4g sodium) – that’s around one teaspoon – and the amount children should eat depends on their age:
| Age | Max. salt / day | Max sodium / day |
|---|---|---|
| 1 to 3 years | 2g salt a day | 0.8g sodium |
| 4 to 6 years | 3g salt a day | 1.2g sodium |
| 7 to 10 years | 5g salt a day | 2g sodium |
| 11 years and over | 6g salt a day | 2.4g sodium |
As a comparison let’s see the sodium content in other foods and liquids:
| Food | Sodium (salt) content |
|---|---|
| Buxton mineral water, 1 litre | 24mg (0.024g) |
| Semi-skimmed milk, 100ml | 100mg (0.1g) |
| Large egg, 50g | 70mg (0.07g) |
| Hovis soft white medium bread, per 100g | 900mg (0.9g) |
| Tesco Fusilli pasta twists, per 100g | 100mg (0.1g) |
| White rice, 100g | 100mg (0.1g) |
| Tesco easy cook brown rice, 100g | 10mg (<0.01g) |
| Tesco chicken breast, 100g | 200mg (0.2g) |
| Tesco boneless cod fillet, 100g | 300mg (0.3g) |
| Carling beer, 100ml | 0mg (0g) |
In Conclusion
Calcium and magnesium are dissolved naturally in water and have many health benefits. However, they can also cause irreparable damage to pipes and appliances at home and in industry. People living in hard water areas therefore often install water softening equipment to prevent this from happening.
This conversion of soft water to hard water undergoes an ion exchange process where calcium and magnesium are replaced with sodium. Higher levels of sodium (salt) raises questions as to whether soft water is safe to drink especially with regard to infants and people with high blood pressure who need to limit their salt intake.
For most people there is no problem drinking softened water – it’s simply a matter of taste and calcium and magnesium are found in a variety of foods and drinks. However, the Drinking Water Inspectorate and water companies do advise installing an additional tap to draw directly from the main supply due to the added sodium.